In recent years, significant progress has been made in the research and development of anti-inflammatory and anti-infective drugs due to the improvement of small molecule drug screening technology and the development of bio-macromolecule drugs such as neutralizing antibodies. In the early stage of drug screening evaluation, it is necessary to select cheap and feasible inflammatory animal models and evaluation methods as the preliminary pharmacodynamic evaluation program.
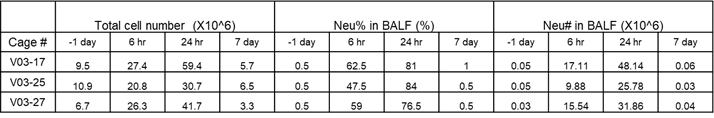
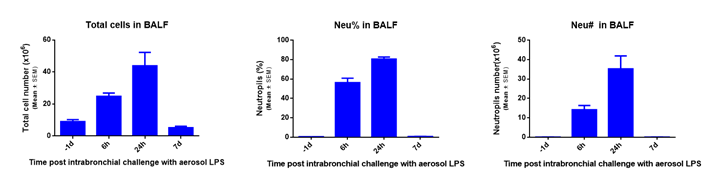
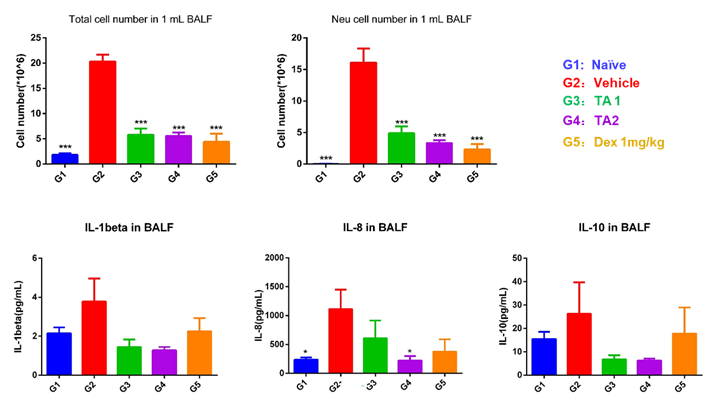
For Lung Inflammation model establishment, we chose the mouse lipopolysaccharide (LPS) induced acute pneumonia model to validate it’s in vivo efficacy evaluation. Methods: LPS-induced acute pneumonia model was established by nasal inhalation method, and the pathological grading standard was established by HE staining of lung injury at different time points.